Low-Cost Generics Giant Sun Pharma Targets Biosimilars With Organon Acquisition
Input
Modified
$11.75 Billion Organon Deal Marks Largest Acquisition in Indian Pharmaceutical History Preparing for a ‘Biosimilar Golden Age’ Amid Intensifying Global Competition More Personnel, More Clinical Trials and Streamlined Reviews Pose Threat to Korean Biotech
India’s largest pharmaceutical company, Sun Pharma, is reshaping the global biotech landscape with its acquisition of U.S. healthcare company Organon. Through the megadeal, Sun Pharma has vaulted into the ranks of the world’s seventh-largest biosimilar companies. The shift is also expected to affect Korea’s biotech industry. Korean companies have led the global biosimilar market on the strength of technology and quality, but if ultra-low-cost biosimilars from India begin spreading in earnest, pressure from price competition will become unavoidable.
A Quantum Leap to Global Top 25 and Seventh in Biosimilars
According to CNBC on the 28th local time, Sun Pharma officially announced its acquisition of Organon the previous day. The total transaction value stands at $11.75 billion, with the boards of both companies approving the deal. The transaction is expected to close in early 2027 after regulatory approvals in the U.S. and India and approval from Organon shareholders. Organon was launched in 2021 after U.S. drugmaker Merck spun off its women’s health products and biosimilar sales business. The company is listed on the New York Stock Exchange and owns more than 70 products, including established branded drugs and biosimilars centered on women’s health, as well as six manufacturing facilities across the European Union and emerging markets.
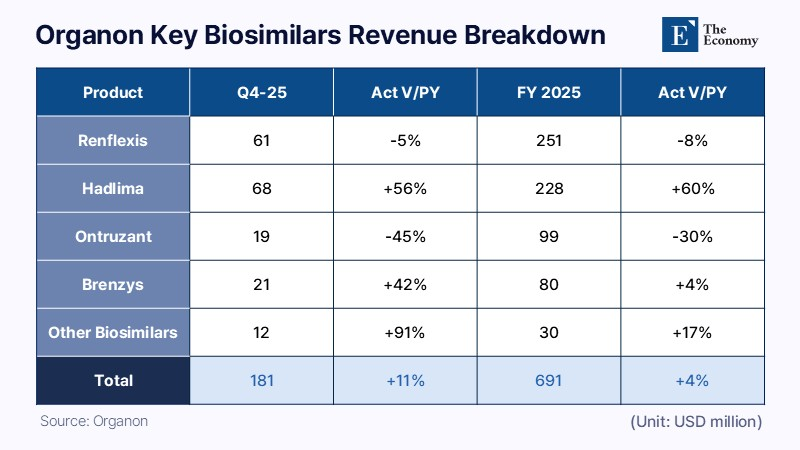
The core of the acquisition lies in Organon’s biosimilar business. Organon’s biosimilar revenue reached $691 million in 2025, up 4% from a year earlier. It accounted for 11% of total revenue of $6.216 billion. Sun Pharma Chairman Dilip Shanghvi said Organon’s portfolio, capabilities and global reach are complementary to Sun Pharma, adding that the combination of the two organizations can create a stronger and more diversified platform.
The transaction is the largest deal in Indian pharmaceutical history and the biggest merger and acquisition in the biopharmaceutical industry this year. Once completed, Sun Pharma is expected to become a global top-25 pharmaceutical company with revenue of $12.4 billion. Its business structure will also change, with innovative medicines expected to account for 27% of revenue after the merger. The company is projected to become a top-three global player in women’s health and a top-seven player in biosimilars. The biosimilar entry effect is particularly significant. Sun Pharma has long demonstrated strengths in generics and specialty generics, but through this deal it will enter the global top 10 in the biosimilar market.
India’s Biosimilar Industry Enters Full Growth Phase as Patent Cliff Meets Industrial Policy
This means Indian pharmaceutical companies have secured the foundational strength needed to compete for leadership on equal footing with Western big pharma. India, a generic-drug powerhouse, has recently been targeting the drug market centered on biosimilars, backed by the Indian government’s bio-manufacturing policy and regulatory overhaul. The Department of Pharmaceuticals under India’s Ministry of Chemicals and Fertilizers has estimated that the revenue scale of drugs entering the patent cliff zone stands at $251 billion, presenting it as a new growth opportunity for India’s pharmaceutical industry.
At the same time, India’s Central Drugs Standard Control Organization revised its biosimilar drug guidelines last year, refining standards for comparability, quality, clinical trials and pharmacovigilance. The Indian government is also moving to strengthen a bio-manufacturing ecosystem centered on biosimilars through the BioE3 policy approved in 2024 and Biopharma SHAKTI announced in 2026. These steps reflect the limits of a growth model centered on generic drugs. The generic market based on synthetic medicines has recently faced declining profitability and slowing growth amid drug-price cuts and intensified competition in major markets including the U.S., Europe and Japan. By contrast, biologics such as antibody therapies and cell and gene therapies are expanding rapidly on the back of rising chronic diseases and shifts in treatment paradigms.
The scale growth of India’s pharmaceutical industry is also supporting the expansion of biosimilars. According to the Indian government, India has the world’s third-largest pharmaceutical production capacity, with annual revenue of $50.405 billion as of FY25. India’s domestic pharmaceutical market is also projected to expand from $60 billion in 2025 to $130 billion in 2030. On the foundation of this large pharmaceutical industry, biosimilars are emerging as a next-generation growth field with higher entry barriers and greater added value than conventional generics.
This growth base is expected to generate synergy when combined with India’s core competitiveness: its price structure. India, which has dominated the global generics market on the back of low production costs, is now seeking to further elevate price competitiveness through policy support for biologics. For example, local company Levim became the first in the world to produce the diabetes treatment biosimilar Liraglutide and is selling it at one-third the price of imported drugs. In that process, the government covered up to 85% of clinical trial costs.
In February, India also unveiled a plan to invest $1.2 billion over five years to strengthen competitiveness in the bio industry. India’s investment may appear small compared with bio-industry fiscal support in the U.S. and Europe, but the difference in cost structure must be taken into account. According to the International Monetary Fund, India’s per-capita gross domestic product stood at $3,050 last year, far below the U.S. at $90,000 and Europe at $50,000. In general, the lower the per-capita GDP, the lower the unit costs for labor, clinical trials and facility operations tend to be. Particularly in research and development and clinical fields, where labor input is high, the same budget can secure more personnel and sites.
Biosimilar Powerhouse Korea on Alert Over India’s Rapid Pursuit
India’s rise is expected to place direct pressure on Korea, a biosimilar powerhouse. According to the Korea Biotechnology Industry Organization, Korea obtained a total of five biosimilar approvals from the U.S. Food and Drug Administration last year. During the same period, the FDA approved sales of 18 biosimilars in total, with Korea accounting for roughly 27.8%, reaffirming its standing. Korea had also ranked joint first with the U.S. in 2024, accounting for four of the 18 approvals, or 22.2%. The U.S., which received four biosimilar approvals from the FDA at the time, slowed somewhat last year with two approvals.
A notable change is India’s sharply rising penetration of the U.S. biosimilar market. By cumulative approvals by country from 2015 through last year, the total 81 approvals were distributed as follows: 28 for the U.S., 19 for Korea, 10 for India, eight for Germany, seven for Switzerland, four for China, two for Iceland, and one each for Taiwan, France and the U.K. India, which had six cumulative approvals through 2024 and ranked joint fourth with Germany, received four biosimilar approvals last year alone, overtaking Germany and Switzerland to rise to third place.
Industry observers expect competition in the U.S., the world’s largest market, to intensify further as the FDA moves this year to simplify biosimilar approval procedures. The outlook is gaining weight as the U.S. shows signs of eliminating Phase 3 comparative efficacy studies. Conducting CES typically requires one to three years and costs a total of $24 million, so its abolition would sharply lower the development threshold. If development costs fall amid already fierce unit-price competition, the competitive front previously concentrated on blockbuster drugs could expand to middle-blockbusters.
Some observers also suggest that, as market entry by latecomers accelerates, securing interchangeable status and building local distribution strategies could emerge as decisive competitive points. If a biosimilar receives FDA approval for interchangeability, which is separate from product approval, it can be substituted at pharmacies for the original drug without a separate physician prescription. One biotech industry official said, “As discussions over abolishing or easing interchangeability continue, competition to obtain that status is also becoming more intense. Yet because the hurdle remains high, it still functions as a core competitive factor in terms of quality credibility,” adding, “Leading companies also maintain advantages over latecomers in local marketing capabilities, and that will serve as an important strength in biosimilar competition.”